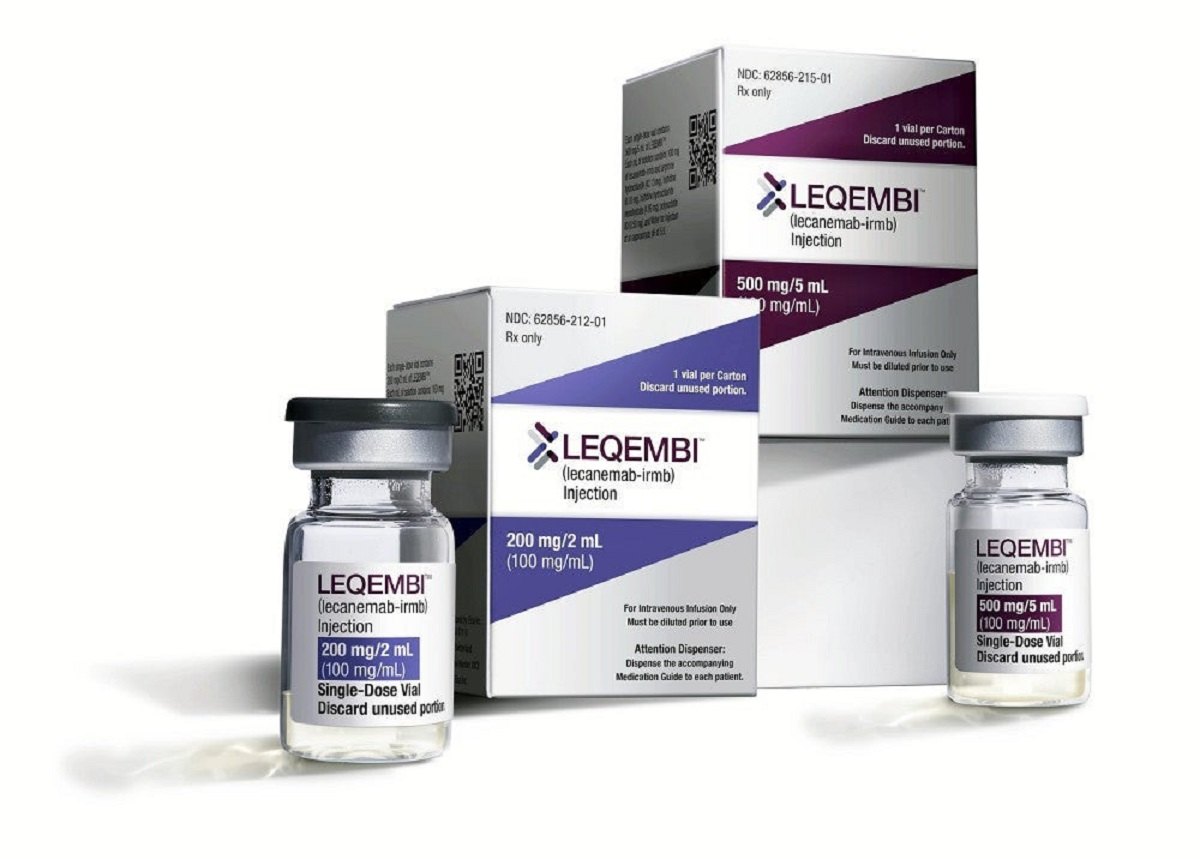
Alzheimer’s drug lecanemab
Reuters
10:39 JST, January 16, 2023
Japanese drugmaker Eisai Co Ltd 4523.T said on Monday it had submitted a marketing authorisation application in Japan for its Alzheimer’s drug lecanemab, which was recently granted accelerated approval in the United States.
The drug, developed in partnership with Biogen Inc BIIB.O, is an antibody that has been shown to remove sticky deposits of a protein called amyloid beta from the brains of those in the early stages of the mind-wasting disease.
The company’s application is based on results from a late-stage study in which the drug was shown to reduce the rate of cognitive decline in patients with early Alzheimer’s by 27%, compared with a placebo.
Most Read
Popular articles in the past 24 hours
-

German Journalist Traces Footsteps of Great-Grandfather Who Was P...
-
10 Foreign Tourists Sustain Injuries After Fire at Capsule Hotel ...
-

World Baseball Classic: Venezuela Beats Japan in Nail-biting Back...
-

Rapid Services Resume on JR Chuo Line; Suspension Caused by Accid...
-

Rapid Services on JR Chuo Line Suspended; Accident at Kokubunji S...
-

World Baseball Classic: Venezuela Beats Japan in Back-and-Forth Q...
-

World Baseball Classic: Red Sox’s Abreu Hits 3-run Homer, Giving ...
-

Zelenskiy Says Ukraine Wants Money, Technology in Return for Midd...
Popular articles in the past week
-

Japan Figure Skating Legend Yuzuru Hanyu Is Proud Disaster Surviv...
-
15 Measles Patients Confirmed in Tokyo in Past 6 Days; 1 May Have...
-

Massive Sewer Pipe Found Jutting Out of Highway in Osaka
-

Japan Govt to Tighten Requirements to Receive Permanent Residency...
-

Power Outage Forces About 980 Passengers in Yokohama to Walk to T...
-

JR Tokai Breaks Ground on Yamanashi Maglev Station; Will Be Part ...
-

Parents in Japan to Get Instagram Notifications When Teens Repeat...
-

'World's Oldest Bio-Business' Is Japan's Seed Koji Retailing, Mol...
Popular articles in the past month
-

Producer Behind Pop Group XG Arrested for Cocaine Possession
-

Japan PM Takaichi’s Cabinet Resigns en Masse
-

Man Infected with Measles Reportedly Dined at Restaurant in Tokyo...
-

Japan Figure Skating Legend Yuzuru Hanyu Is Proud Disaster Surviv...
-

iPS Treatments Pass Key Milestone, but Broader Applications Far f...
-

Sanae Takaichi Elected Prime Minister of Japan; Keeps All Cabinet...
-

South Korea Tightens Rules on Foreigners Buying Homes in Seoul Me...
-

Nepal Bus Crash Kills 19 People, Injures 25 Including One Japanes...
Top Articles in Business
-

Nippon Life Insurance’s U.S. Arm Sues OpenAI Over Legal Assistance Provided by ChatGPT
-

Japan, U.S. Name 3 Inaugural Investment Projects; Reached Agreement After Considerable Difficulty
-

Japan’s Major Real Estate Firms Expanding Overseas Businesses to Secure Future Growth, Focusing on Europe, U.S., Asia
-

JR Tokai Breaks Ground on Yamanashi Maglev Station; Will Be Part of Linear Chuo Shinkansen Line from Tokyo to Nagoya
-

Transport Companies See Opportunity in Narita Expansion; Airlines, Railways Prepare to Meet Expected Growth in Demand
JN ACCESS RANKING
-

Producer Behind Pop Group XG Arrested for Cocaine Possession
-

Japan PM Takaichi’s Cabinet Resigns en Masse
-

Man Infected with Measles Reportedly Dined at Restaurant in Tokyo Station
-

Japan Figure Skating Legend Yuzuru Hanyu Is Proud Disaster Survivor and Gold Medalist, Vows to Continue Support Efforts
-

iPS Treatments Pass Key Milestone, but Broader Applications Far from Guaranteed